Alpha Synuclein Oligomers: Dopamine Stabilization and Neurodegeneration
What is an oligomer?
An oligomeric protein consists of few repeating units, as opposed to a polymer, which has many units, or monomer, which has only one unit. Oligo– is derived from the Ancient Greek word meaning “few” and mer comes from the Ancient Greek word for “part.”
Alpha synuclein is a protein implicated in Parkinson’s Disease (PD), a common neurodegenerative disorder. While alpha synuclein is normally found in the presynaptic terminals of neurons, in certain diseases, known as synucleinopathies, it aggregates to form larger fibrils and aggregates called Lewy bodies. During this aggregation process, it forms intermediate species called oligomers. These oligomers are of interest because they, rather than Lewy bodies, are increasingly thought to be the toxic species that cause neuron death in synucleinopathic diseases.1,2
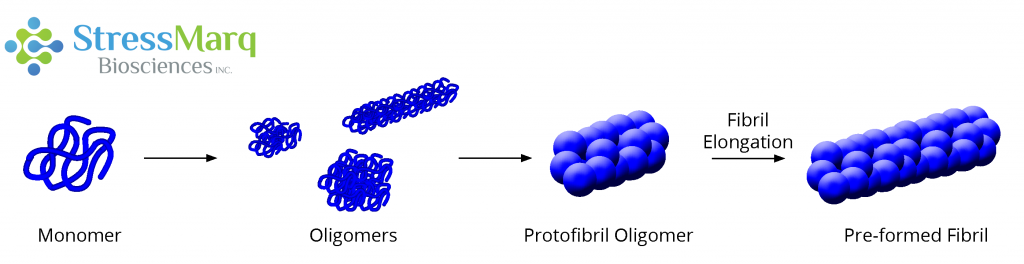
Alpha synuclein monomers can aggregate to form a variety of oligomeric species. If aggregation continues, they form protofibrils and fibrils as found in Lewy bodies.
How Dopamine Induces Alpha Synuclein Oligomerization
Dopamine is a neurotransmitter that can inhibit the aggregation of alpha synuclein into fibrils, stabilizing it in its oligomeric form. Dopamine oxidation is involved in this process.
Dopamine Oxidation Mechanism
The first step in dopamine oxidation is the oxidation of the catechol ring to form dopamine-quinone. This species can then react to form dopaminochrome, which then can be converted into 5, 6-indoloquinone and ultimately neuromelanin.3
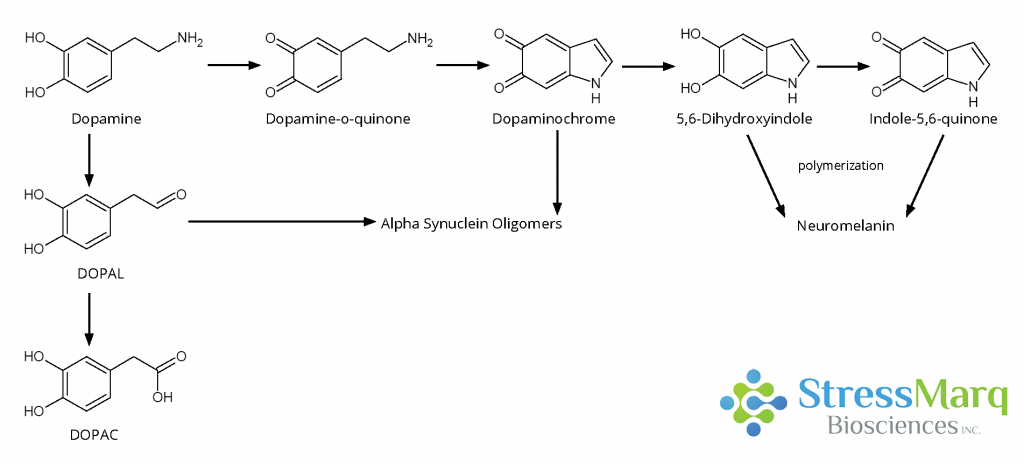
Dopamine oxidation mechanism. Both dopaminochrome and DOPAL are thought to interact with alpha synuclein in a way that promotes oligomer formation
Interactions with Alpha Synuclein: Oligomer Stabilization
Dopamine and its derivatives are thought to inhibit alpha synuclein aggregation and kinetically stabilize oligomers through interactions in different regions:
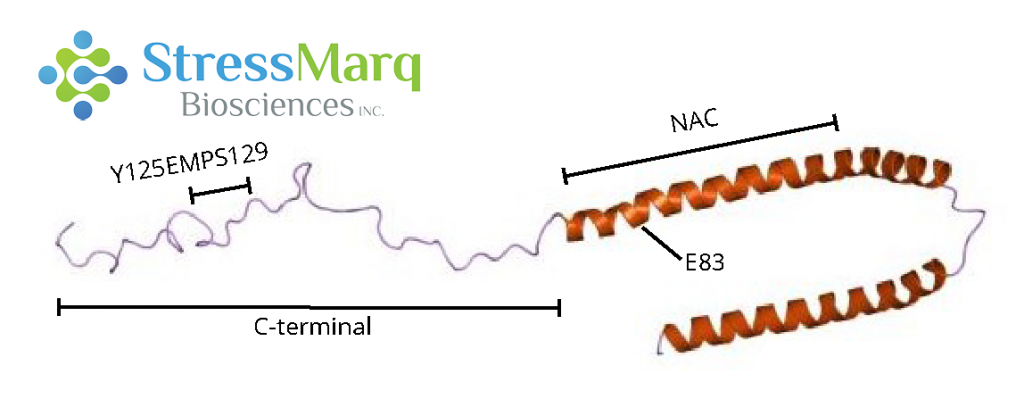
C-terminus: Y125EMPS129
Computational modelling indicates that the aromatic ring of dopamine interacts with this 5-amino acid sequence of alpha synuclein in a non-specific, non-covalent, hydrophobic manner.4 However, there is also evidence that the oxidation of the methionine residue in this region, and not binding with dopamine, that inhibits fibrillization.5 In any case, this motif is required for oligomer stabilization6-9 and the mutation of Y125EMPS129 to F125AAFA129 prevents this by preventing interaction with dopamine.7,11
NAC Region: E83
Dopamine derivatives are thought to also have long-range electrostatic interactions with E83 in the NAC region.4 The mutation E83A impairs dopamine’s ability to inhibit fibril formation.4
Oligomerization Mechanism
Dopamine and its derivatives not only kinetically stabilize oligomers, but can also induce oligomerization of monomers. Dopamine can induce dimerization, with residues 43 through 60 in the N-terminal forming the dimer interface.10 Dopamine also induces trimerization, where monomers overlap but do not have associate end-to-end.11 The oxidation of four methionine residues at the C and N-terminals prevents end-to-end associations and beta-sheet formation by cross-linking with dopamine-quinone and dopamine-melanin.11 Dopaminochrome can induce reversible conformational changes that promote oligomer formation12 and DOPAL, a toxic dopamine metabolite, is thought to promote monomer aggregation by cross-linking alpha synuclein lysine residues.13
Alpha Synuclein Oligomer Structure
There are many varieties of alpha synuclein oligomers, both found in living systems and synthesized in labs. Alpha synuclein oligomers derived from post-mortem brains can range from dimers (two units) to 150 units.1,14
Evidence for Oligomer Toxicity
Alpha synuclein oligomers are increasingly recognized as a toxic species in synucleinopathies. In vitro experiments and animal models have demonstrated their toxicity, as cell death and degeneration occurs when there are oligomers present, even in the absence of large alpha synuclein aggregates.15-18
However, there are also non-toxic varieties of oligomers. The most toxic oligomers are thought to be between 2 and 6 nm in diameter,14 whereas larger oligomers are thought to be non-toxic but capable of seeding.1
Alpha Synuclein Oligomer Toxicity Mechanisms
Alpha synuclein oligomers can be toxic to cells through various mechanisms:
Interactions with Lipid Membranes and Pore Formation
Alpha synuclein oligomers have exposed hydrophobic surfaces19 and accessible N-terminal regions that enable them to interact with and destabilize lipid membranes.20 Small, annular oligomers are likely to form pores in vesicle and plasma membranes.21-25 Oligomer hydrophobicity is thought to be associated with toxicity since hydrophobic oligomers can bind more tightly to membranes and organelles.26 This would account for the decreased toxicity in larger aggregates with fewer accessible hydrophobic patches.
Calcium Influx
Alpha synuclein oligomers can induce Ca2+ influx via N-type voltage-dependent Ca2+.27 This process is thought to be mediated by the NAC region.28 While alpha synuclein monomers can also affect calcium signalling, only oligomers induce cell death.29
Mitochondrial Dysfunction
Alpha synuclein oligomers can cause mitochondrial dysfunction by inducing a slow and progressive increase in NADH.30 This is associated with permeability transition pore (PTP) opening, increased production of reactive oxygen species, inhibition of complex I, mitochondrial depolarization, and reduced electron flow through the electron transport chain.30
Endoplasmic Reticulum (ER) Stress
Alpha synuclein oligomers can form within the ER lumen and accumulate within the ER, increasing neuronal sensitivity to ER stress.31 This ER stress contributes to neurodegeneration.
Glutamatergic Receptors
Globular oligomers can alter the function of glutamatergic receptors, which leads to disrupted neuronal function.31,32
SNARE Complex Inhibition
Large intracellular dopamine-stabilized alpha synuclein oligomers can inhibit the formation of the SNARE complex, which reduces the release of neurotransmitters.33
Alpha Synuclein Oligomer Seeding
Different varieties of oligomers show different seeding capacities. Oligomers with higher seeding capacities are associated with lower toxicities.34 Intracellular alpha synuclein oligomers can be released via exosomes, where they are more likely to be taken up by neighbouring cells than free extracellular alpha synuclein oligomers.35
Dopamine-Stabilized Alpha Synuclein Oligomers
Dopamine-induced oligomers have been shown to auto-aggregate36and can cross-seed amyloid-beta aggregates.37 Dopamine-induced oligomers have also induced neurodegeneration in worm and mouse in vivo models.38 However, it is worth noting that different experimental conditions produce different types of oligomers, and the toxicity is dependent on oligomer synthesis method and structure.
StressMarq manufactures commercially available dopamine-stabilized oligomers for alpha synuclein research. Oligomers are of interest to researchers because they show potential as a biomarker, since they are present in plasma and CSF, as well as a treatment target since they are a cause of neurodegeneration.31
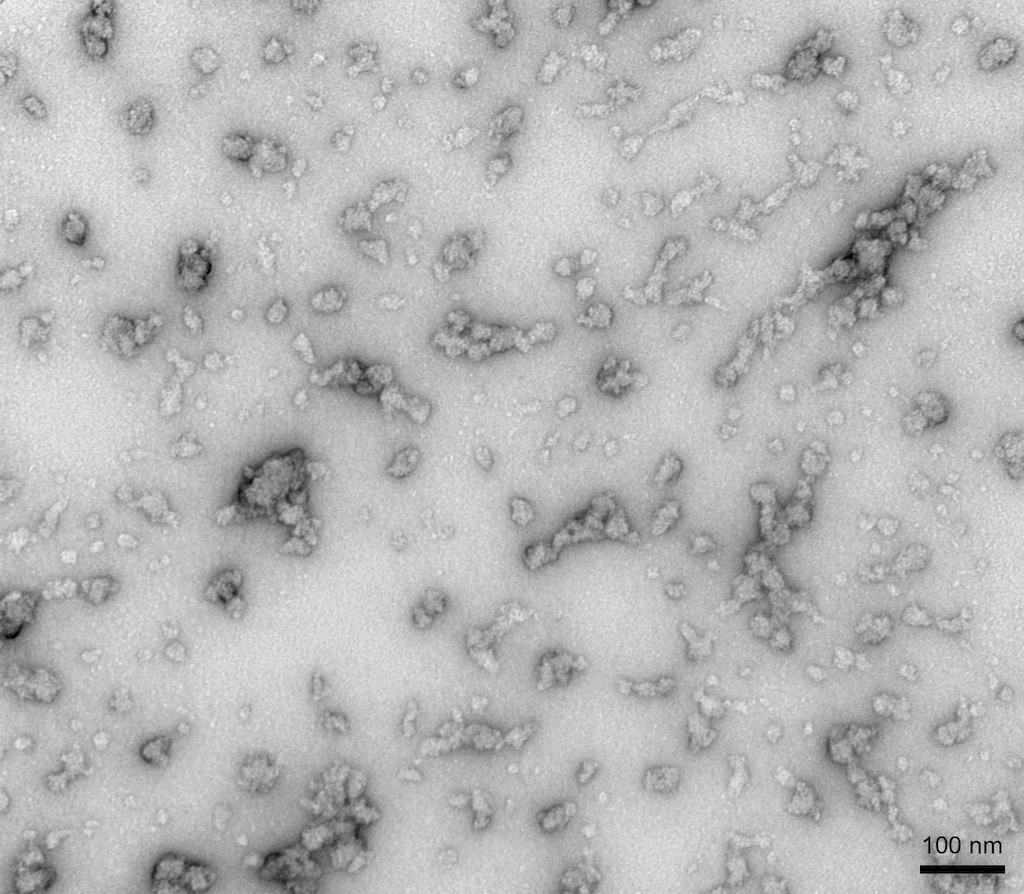
TEM of Human Recombinant Alpha Synuclein Oligomers (Dopamine HCL Stabilized) (SPR-466)
References
- Danzer, K.M., Haasen, D., Karow, A.R., Moussaud, S., Habeck, M., Giese, A., Kretzschmar, H., Hengerer, B., Kotska, M. Different species of alpha-synuclein oligomers induce calcium influx and seeding. J Neurosci. 2007; 27:9220–9232.
- Wright, J.A., Wang, X., Brown, D.R. Unique copper-induced oligomers mediate alpha-synuclein toxicity. FASEB J. 2009; 23(8):2384-93.
- Van Diggelen, F., Tepper, A.W.J.W., Apetri, M.M., Otzen, D.E. α-Synuclein oligomers: a study in diversity. Israel J Chem. 2016; 57(7).
- Herrera, F.E., Chesi, A., Paleologou, K.E., Schmid, A., Muniz, M., Vendruscolo, M., Gustincich, S., Lashuel, H.A., Carloni, P. Inhibition of a-Synuclein Fibrillization by Dopamine Is Mediated by Interactions with Five C-Terminal Residues and with E83 in the NAC Region. PLoS ONE. 2008; 3(10):e3394.
- Leong, S.L., Pham C.L., Galatis, D., Fodero-Tavoletti, M.T., Perez, K., Hill, A.F., et al. Formation of dopamine-mediated alpha-synuclein-soluble oligomers requires methionine oxidation. Free Rad Biol Med. 2009; 46(10):1328–37.
- Mazzulli, J.R., Mishizen, A.J., Giasson, B.I. Cytosolic catechols inhibit alpha-synuclein aggregation and facilitate the formation of intracellular soluble oligomeric intermediates. J Neurosci. 2006; 26:10068–10078.
- Mazzulli, J.R., Armakola, M., Dumoulin, M., Parastatidis, I., Ischiropoulos, H. Cellular oligomerization of alpha-synuclein is determined by the interaction of oxidized catechols with a C-terminal sequence. J Biol Chem. 2007; 282:31621–31630.
- Conway, K.A., Rochet, J.C., Bieganski, R.M., Lansbury, P.T. Kinetic stabilization of the alpha-synuclein protofibril by a dopamine-alpha-synuclein adduct. Science. 2001; 294:1346–1349.
- Norris, E.H., Giasson, B.I., Hodara, R. Reversible inhibition of alpha-synuclein fibrillization by dopaminochrome-mediated conformational alterations. J Biol Chem. 2005; 280:21212–21219.
- Leong, S.L., Hinds, M.G., Connor, A.R., Smith, D.P., Illes-Toth, E., Pham, C.L., Barnham, K.J., Cappai, R. The N-terminal residues 43 to 60 form the interface for dopamine mediated α-synuclein dimerisation. PLoS One. 2015; 10(2):e0116497.
- Rekas, A., Knott, R.B., Sokolova, A., Barnham, K.J., Perez, K.A., Masters, C.L., Drew, S.C., Cappai, R., Curtain, C.C., Pham, C.L. The structure of dopamine induced alpha-synuclein oligomers. Eur Biophys J. 2010; 39(10):1407-19.
- Norris, E.H., Giasson, B.I., Ischiropoulos, H., Lee, V.M. Effects of oxidative and nitrative challenges on alpha-synuclein fibrillogenesis involve distinct mechanisms of protein modifications. J Biol Chem. 2003; 278(29):27230-40.
- Werner-Allen, J.W., Dumond, J.F., Levine, R.L., Bax, A. Toxic Dopamine Metabolite DOPAL Forms an Unexpected Dicatechol Pyrrole Adduct with Lysines of α-Synuclein. Angew Chem Int Ed Eng. 2016; 55(26):7374-8.
- Cremades N, et al. Direct Observation of the Interconversion of Normal and Toxic Forms of alpha-Synuclein. Cell. 2012; 149:1048–1059
- Lee, H.J., Bae, E.J., Lee, S.J. Extracellular α–synuclein-a novel and crucial factor in Lewy body diseases. Nat Rev Neurol. 2014; 10(2):92-8.
- Xu, J., Kao, S.Y., Lee, F.J., Song, W., Jin, L.W., Yanker, B.A. Dopamine-dependent neurotoxicity of alpha-synuclein: a mechanism for selective neurodegeneration in Parkinson disease. Nat Med. 2002; 8(6):600-6.
- Van der Putten, H., Wiederhold, K.H., Probst, A., Barbieri, S., Mistl, C., Danner, S., Kauffman, S., Hofele, K., Spooren, W.P., Ruegg, M.A., Lin, S., Caroni, P., Sommer, B., Tolnay, M., Bilbe, G. Neuropathology in mice expressing human alpha-synuclein. J Neurosci. 2000; 20(16):6021-9.
- Lo Bianco, C., Ridet, J.L., Schneider, B.L., Deglon, N., Aebischer, P. alpha -Synucleinopathy and selective dopaminergic neuron loss in a rat lentiviral-based model of Parkinson’s disease. Proc Natl Acad Sci USA. 2002; 99(16):10813-3.
- Van Rooijen, B.D., Claessens, M.M.A.E., Subramaniam, V. Lipid bilayer disruption by oligomeric α-synuclein depends on bilayer charge and accessibility of the hydrophobic core. Biochim Biophys Acta Biomem. 2009; 1788(6):1271-1278.
- Fusco, G., Chen, S.W., Williamson, P.T.F., Cascella, R., Perni, M., Jarvis, J.A., Cecchi, C., Vendruscolo, M., Chiti, F., Cremades, N., Ying, L., Dobson, C.M., De Simone, A. Structural basis of membrane disruption and cellular toxicity by α-synuclein oligomers. 2017; 358(3639):1440-1443.
- Volles, M.J., Lee, S.J., Rochet, J.C. Vesicle permeabilization by protofibrillar alpha-synuclein: implications for the pathogenesis and treatment of Parkinson’s Disease. Biochem. 2001; 40:7812-7819.
- Ding, T.T., Lee, S.J., Rochet, J.C., Lansbury, P.T. Annular alpha-synuclein protofibrils are produced when spherical protofibrils are incubated in solution or bound to brain-derived membranes. Biochem. 2002; 41:10209-10217.
- Gosavi, N., Lee, H.J., Lee, J.S., Patel, S., Lee, S.J. Golgi Fragmentation Occurs in the Cells with Prefibrillar α-Synuclein Aggregates and Precedes the Formation of Fibrillar Inclusion. J Biol Chem. 2002; 277:48984-48992.
- Lashuel, H.A., Petre, B.M., Wall, J., Simon, M., Nowak, R.J., Walz, T., Lansbury, P.T. Alpha-synuclein, especially the Parkinson’s disease-associated mutants, forms pore-like annular and tubular protofibrils. J Mol Biol. 2002; 322(5):1089-102.
- Mosharov, E.V., Staal, R.G., Bove, J., Prou, D., Hananiya, A., Markov, D., Poulsen, N., Larsen, K.E., Moore, C.M., Troyer, M.D., Edwards, R.H., Przedborski, S., Sulzer, D. Alpha-synuclein overexpression increases cytosolic catecholamine concentration. J Neurosci. 2006; 26(36):9304-11.
- Lee, J.E., Sang, J.C., Rodrigues, M., Carr, A.R., Horrocks, M.H., De, S. Bongiovanni, M.N., Flagmeier, P., Dobson, C.M., Wales, D.J., Lee, S.F., Klenerman, D. Mapping Surface Hydrophobicity of α-Synuclein Oligomers at the Nanoscale. Nano Lett. 2018; 18(12):7497-7501.
- Adamczyk, A., Strosznajder, J.B. Alpha-synuclein potentiates Ca2+ influx through voltage-dependent Ca2+ channels. Neuroreport. 2006; 17(18):1883-6.
- Butler, B., Sambo, D., Khoshbouei, H. Alpha-synuclein modulates dopamine neurotransmission. J Chem Neuroanat. 2017; 83-84:41-49.
- Angelova, P.R., Ludtmann, M.H., Horrocks, M.H., Negoda, A., Cremades, N., Klenerman, D., Dobson, C.M., Wood, N.W., Pavlov, E.V., Gandhi, S., Abramov, A.Y. Ca2+ is a key factor in α-synuclein-induced neurotoxicity. J Cell Sci. 2016; 129(9):1792-801.
- Ludtmann, M.H.R., Angelova, P.R., Horrocks, M.H., Choi, M.L., Rodrigues, M., Baev, A.Y., Berezhnov, A.V., Yao, Z., Little, D., Banushi, B., Al-Menhali, A.S., Ranasinghe, R.T., Whiten, D.R., Ratsuda, Y. Dolt, K.S., Devine, M.J., Gissen, P. Kunath, T., Jaganjac, M., Pavlov, E.V., Klenerman, D., Abramov, A.Y., Gandhi, S. α-synuclein oligomers interact with ATP synthase and open the permeability transition pore in Parkinson’s disease. Nat Commun. 2018; 9:2293.
- Kalia, L.V., Kalia, S.K., McLean, P.J., Lozano, A.M., Lang, A.E. α-Synuclein oligomers and clinical implications for Parkinson disease. Ann Neurol. 2013; 73(2):155-169.
- Diogenes, M.J., Dias, R.B., Rombo, D.M., Vincente Miranda, H., Maiolino, F., Guerreiro, P., Nasstrom, T., Franquelim, H.G., Oliveira, L.M., Castanho, M.A., Lannfelt, L., Bergstrom, J., Ingelsson, M., Quintas, A., Sebastiao, A.M., Lopes, L.V., Outeiro, T.F. Extracellular alpha-synuclein oligomers modulate synaptic transmission and impair LTP via NMDA-receptor activation. J Neurosci. 2012; 32(34):11750-62.
- Choi, B.K., Choi, M.G., Kim, J.Y., et al. Large alpha-synuclein oligomers inhibit neuronal SNARE-mediated vesicle docking. Proc Natl Acad Sci USA. 2013; 110:4087–4092.
- Mor, D.E., Ischiropoulos, H. The Convergence of Dopamine and α-Synuclein: Implications for Parkinson’s Disease. J Exp Neurosci. 2018; 20(11):1560-1568.
- Danzer, K.M., Kranich, L.R., Ruf, W.P., Cagsal-Getkin, O., Winslow, A.R., Zhu, L., Vanderburg, C.R., McLean, P.J. Exosomal cell-to-cell transmission of alpha synuclein oligomers. Molecular Neurodegeneration. 2012; 7:42.
- Pieri, L, Madiona, K., Melki, R. Structural and functional properties of prefibrillar α-synuclein oligomers. Sci Rep. 2016; 6:24526.
- Planchard, M.S., Exley, S.E., Morgan, S.E., Rangachari, V. Dopamine-induced α-synuclein oligomers show self- and cross-propagation properties. Protein Sci. 2014; 23(10):1369-79.
- Mor, D.E., Tsika, E., Mazzulli, J.R., Gould, N.S., Kim, H., Daniels, M.J., Doshi, S., Gupta, P., Grossman, J.L., Tan, V.S., Kalb, R.G., Caldwell, K.A., Caldwell, G.A., Wolfe, J.H., Ischiropoulos, H. Dopamine induces soluble α-synuclein oligomers and nigrostriatal degeneration. Nat Neurosci. 2017; 20(11):1560-1568.


Leave a Reply