Parkinson’s Disease Models: An Overview of Cellular and Animal Models for Neurodegeneration Research
Disease Models
For centuries, animals have been used in biological research and helped scientists understand the mysteries of life. Experiments on animals have enhanced understanding of anatomy, physiology and pathology and led to great discoveries in medicine1. Parkinson’s Disease (PD) is a common neurodegenerative disorder that is becoming more prevalent with aging populations. Because PD is a complex, heterogeneous disease with diverse clinical and pathological features, PD research involves a variety of disease models.
Disease models are systems, such as cells or animals, that display features seen in the actual disease. For instance, a disease model for PD could be a cell line or animal that includes some of the pathological features of PD, such as alpha synuclein aggregation or dopaminergic neuron degeneration.
Why are disease models important?
Disease models allow for study and experimentation that cannot be done on humans. This helps researchers to better understand how a disease develops as well as to test the efficacy of therapeutic candidates. In drug development, drugs are tested in pre-clinical disease models before they can proceed to clinical trials. Currently, transgenic animals are used extensively as models for human diseases and drug development.
What is a transgenic animal?
Transgenic animals are genetically engineered to have foreign genes inserted into their genomes. This enables researchers to study human genes and diseases in animal (often rodent) models.
What makes a good disease model?
In humans, a disease may take years to progress. Model organisms that generate the disease quickly allow scientists to study it and test potential treatments in shorter time periods. The ideal model would be an organism that is easy to monitor and manipulate in the lab, has low cost and can display the pathophysiology of the disease.
The role of disease models in Parkinson’s Disease
Disease models are used extensively in the study of neurodegenerative disorders and have enhanced our understanding of the molecular mechanisms involved in their progress. In PD research, scientists use animal and cell models to understand the cause and the progression of the disease and discover possible treatments. An ideal model for PD would mimic its pathology, which includes alpha synuclein and Lewy body formation and neuronal loss in the substantia nigra pars compacta, as well as the behavioral symptoms of the disease. However, animal models usually fail in reproducing PD pathophysiology and results from animal model studies do not often translate to humans2,3.
Cellular Models of Parkinson’s Disease
Cell cultures like primary dopaminergic neurons, mesencephalic slice cultures and immortalized cell lines are used as models in PD research. Compared to animal models, cellular models are advantageous because they generate pathology quickly, are inexpensive, and do not have the same ethical concerns involved with animal models. They can reproduce dopaminergic neuron degeneration and alpha synuclein aggregation but usually results from cultured cells ultimately need to be validated in animals. The in vitro models provide useful information on the PD pathogenesis and are useful tools in drug development4,5.
Stem Cell Models of Parkinson’s Disease
Advances in stem cell technology have provided neurodegenerative research with useful tools for disease modeling and drug screening. Recent studies used human induced pluripotent stem cells (iPSCs) to model PD. iPSCs can be generated directly from somatic cells of patients and they are differentiated into midbrain dopaminergic neurons6. Neurons derived from iPSCs of patients can show how mutations on a gene generate PD phenotypes7.
Animal Models of Parkinson’s Disease
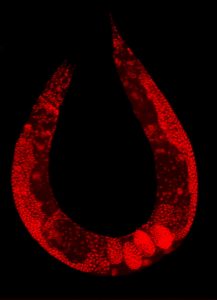
Wild-type C. elegans hermaphrodite stained to highlight the nuclei of all cells.
A wide range of living organisms have been used in PD research to investigate the pathogenesis of this neurodegenerative disorder. There are four animal groups that are commonly used: rodents, non-human primates, other mammalian and non-mammalian species. Rats and mice are the most popular and widely used for modelling PD because of their small size, low cost, ease of handling, and similar anatomy to humans. Nonhuman primate models and other mammalian species like monkeys, dogs and cats are rarely used because of the cost and ethical concerns. Non-mammalian species like Drosophila melanogaster (fruit fly), Danio rerio (zebrafish) and Caenorhabditis elegans (nematode) have been widely used to study the molecular mechanisms of PD3. They are more simple organisms that are low-cost and easy to manipulate genetically, but their short lifespan and simple anatomy make it difficult to model the complex mechanisms of PD8,9.
Animal models of PD can be divided into two categories: neurotoxin and genetic models.
Neurotoxin Models of Parkinson’s Disease
Neurotoxin- based models produced by 6-hydroxydopamine (6-OHDA), 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), rotenone and paraquat are widely used because of their ability to cause oxidative stress that leads to dopaminergic neuronal death10. These models have been very useful and valuable tools as they are easily made and mimic the symptoms of PD like motor deficits, but do not produce its pathological features. The main drawback of this model is that induces rapid neurodegeneration causing motor deficits which is contradictory to the natural progression of the disease in humans11. PD is a chronic neurodegenerative disorder and the degeneration of the dopaminergic neurons in the substantia nigra can start years before the first motor symptoms develop.
Genetic Models of Parkinson’s Disease
Familial forms of PD can be caused by mutations in the genes SNCA, LRRK2, PINK1 and DJ1. The discovery of the alpha synuclein and parkin genes has shown that genetic mutations lead to the development of PD phenotypes13. Transgenic animal models carrying mutations of these genes have been developed to investigate PD but many of these models lack dopaminergic neurodegeneration or cannot display regular motor deficits. Transgenic mice overexpressing A53T mutant alpha synuclein is an exception to these models as they develop motor deficits and synucleinopathy20. Studies have shown that not only genetic but also environmental factors are significant in the PD progression. Combining genetic and toxicity models by exposing a transgenic animal to a toxin could provide a model with both motor deficits and dopaminergic neuron degeneration9.
Alpha Synuclein-based Animal Models
After the discovery that the aggregated and misfolded alpha synuclein plays crucial role in the pathogenesis of PD, new animal models have been developed that reproduce pathological features of the disease. Many transgenic mice overexpressing wild type or mutant alpha synuclein have been generated for PD research.
-
Viral Vector-Based Alpha Synuclein Model
Viral vectors are viruses able to transfer genes into cells. They can be used for overexpression or silencing of certain genes. Recombinant adeno-associated viral (AAV) vectors are used to express human wild-type or mutated alpha synuclein selectively in dopamine neurons which can trigger PD pathology14. This model offers the opportunity to study PD development and the neurodegenerative changes in midbrain dopaminergic neurons. One drawback of using viral vectors is that the levels of expressed alpha synuclein are higher when compared to PD patients15. Another disadvantage of the AAV alpha synuclein model is that the pathology spreading is limited to the nigrostriatal system compared to preformed fibrils which cause wide spreading of the pathology16.
-
Alpha Synuclein Preformed Fibrils (PFFs) Model
In the last several years, researchers developed a new model focusing on aggregated forms of alpha synuclein found in Lewy bodies, the major hallmark of PD. In this model, recombinant alpha synuclein monomers are incubated to generate alpha synuclein preformed fibrils which have similar structure to the alpha synuclein in Lewy bodies and Lewy neurites. These PFFs are then sonicated to generate shorter fibrils and after they are injected into mice. In the neurons the PFFs act as seeds for alpha synuclein aggregates and induce Lewy body pathology. Adding alpha synuclein fibrils to cells or animals causes the monomeric alpha synuclein, which is already present in neurons, to become phosphorylated and aggregate. This pathology can spread to the opposite side of the brain to the injection site17.
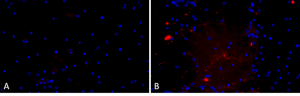
Primary rat hippocampal neurons show lewy body inclusion formation when treated with A53T mutant Alpha Synuclein Protein Preformed Fibrils (SPR-326) (B) but not when treated with a media control (A).
This model is becoming increasingly popular as it can be used in both cell cultures and animals. Compared to the viral vector–based model, alpha synuclein levels are closer to that of the human condition as the pathology is induced in normal levels of the protein. One weakness of this model is that there are variabilities of results due to different practices and improper preparation of the preformed fibrils can lead to a failure in pathogenicity. Sonication of PFFs is required and seeding capacity can be affected by sonicator type and settings16.
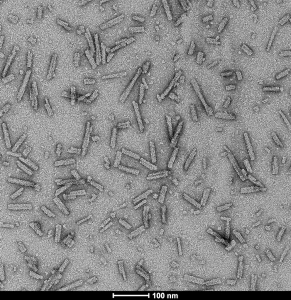
TEM of A53T alpha synuclein preformed fibrils (SPR-326).
A new model that has emerged is a combination of human alpha synuclein preformed fibrils and AAV-mediated overexpression of human alpha synuclein18. This model reproduces many of the PD features in a short time which makes it promising in future studies. Another popular model that many research groups use is the alpha synuclein PFFs injected in transgenic mice that express wild-type and mutant alpha synuclein. In transgenic mice overexpressing human A53T mutant alpha synuclein, the intracerebral injection of alpha synuclein PFFs accelerates the neurological symptoms and death19.
StressMarq Biosciences is the only reagent company worldwide to provide high quality alpha synuclein preformed fibrils. Type 1 human and mouse PFFs and monomers have all been tested in vivo and generate alpha synuclein pathology in rat brains within 30 days of injection. All lots are tested for seeding capacity using Thioflavin T and fibril structure is confirmed using TEM.
REFERENCES
- A Brief History of Animal Modeling, Ericsson A. et al (2013) Mo Med. 110(3): 201–205.
- Limitations of Animal Models of Parkinson’s Disease, Potashkin J. et al. Parkinson’s Dis. 2011:658083
- The benefits and limitations of animal models for translational research in neurodegenerative diseases, Jucker M (2010). Nat. Med. 16, 1210–1214
- Cellular models for Parkinson’s disease, Falkenburger B. et al. (2016) J Neurochem 139:121-130
- Limitations of cellular models in Parkinson’s disease research, Falkenburger B. et al (2006) J Neural Transm 70:261-268
- Opportunities and challenges for the use of induced pluripotent stem cells in modelling neurodegenerative disease, Wu Y. et al. (2019) Open Biol. 9:180177.
- Modeling Parkinson’s Disease using patient-specific induced pluripotent stem cells, Li H. et al. (2018) J Parkinson’s Dis 8:479–493
- Animal models for Parkinson’s Disease Research: Trends in the 2000s, Kin K. et al. (2019) Int J Mol Sci. 2019 Nov; 20(21): 5402.
- Evaluation of models of Parkinson’s disease, Jagmag S. et al. (2016). Front. Neurosci. 9:503.
- Toxin-Induced Models of Parkinson’s Disease, Bové J. et al. (2005) NeuroRx 2(3):484-494
- Neurotoxin-Induced Animal Models of Parkinson Disease: Pathogenic Mechanism and Assessment, Zeng XS. et al. (2018) ASN Neuro. 10:1759091418777438.
- Animal models of Parkinson’s disease, Konnova E. et al. (2018) Parkinson’s Disease: Pathogenesis and Clinical aspects, Chapter 5.
- Genetic models of Parkinson’s disease, Lim KL. et al. (2008) Biochim Biophys Acta. 1792(7):604-15
- Viral Vector-Based Modeling of Neurodegenerative Disorders: Parkinson’s Disease, Fischer L. et al. (2016) Methods Mol Biol. 1382:367-382
- Viral Vector-Based Models of Parkinson’s disease, Van der Perren A. et al (2014) Current topics in behavioral neurosciences22 (2014): 271-301.
- Best Practices for Generating and Using Alpha-Synuclein Pre-Formed Fibrils to Model Parkinson’s Disease in Rodents, Polinski N. et al. (2018) J Parkinsons Dis. 8(2):303–322
- Intracerebral inoculation of pathological α-synuclein initiates a rapidly progressive neurodegenerative α-synucleinopathy in mice, Luk K. (2012) JEM 209(5):975-986
- Modeling Parkinson’s disease pathology by combination of fibril seeds and α-synuclein overexpression in the rat brain, Thakur P. (2017) PNAs 114(39): E8284-E8293
- Pathological a-Synuclein transmission initiates Parkinson-like neurodegeneration in nontransgenic mice, Luk K. et al (2012) Science 338:949
- Giasson, B. I. et al. (2002) ‘Neuronal alpha-synucleinopathy with severe movement disorder in mice expressing A53T human alpha-synuclein.’, Neuron, 34(4), pp. 521–33. doi: 10.1016/s0896-6273(02)00682-7


“Transgenic mice overexpressing A53T mutant alpha synuclein is an exception to these models as they develop motor deficits and synucleinopathy.”
– should be cited:
Giasson, B. I. et al. (2002) ‘Neuronal alpha-synucleinopathy with severe movement disorder in mice expressing A53T human alpha-synuclein.’, Neuron, 34(4), pp. 521–33. doi: 10.1016/s0896-6273(02)00682-7.
Thank you very much for alerting us to this error. I have added the citation to the article and apologize that this was neglected in the original write-up.