Properties
| Storage Buffer | PBS pH 7.4, 50% glycerol, 0.09% Sodium azide *Storage buffer may change when conjugated |
| Storage Temperature | -20ºC, Conjugated antibodies should be stored according to the product label |
| Shipping Temperature | Blue Ice or 4ºC |
| Purification | Protein G Purified |
| Clonality | Monoclonal |
| Clone Number | 6F10 |
| Isotype | IgG2b |
| Specificity | Specific for 4-Hydroxy-2-hexenal (4-HHE) and 4-Hydroxynonenal (4-HNE) modified proteins. Does not detect free 4-Hydroxy-2-hexenal (4-HHE) or 4-Hydroxynonenal (4-HNE). Does not cross-react with Acrolein, Crotonaldehyde, Hexanoyl Lysine, Malondialdehyde, or Methylglyoxal modified proteins. |
| Cite This Product | StressMarq Biosciences Cat# SMC-510, RRID: AB_2702819 |
| Certificate of Analysis | A 1:1000 dilution of SMC-510 was sufficient for detection of 4-Hydroxy-2-hexenal in 0.5 µg of 4-Hydroxy-2-hexenal conjugated to BSA and 4-Hydroxy nonenal conjugated to BSA by ECL immunoblot analysis using Goat Anti-Mouse IgG:HRP as the secondary Antibody. |
Biological Description
| Alternative Names | 4-Hydroxy-2-hexenal (4-HHE) Antibody, 4-Hydroxy-2-hexenal Antibody, 4-HHE Antibody, MG Antibody, MG-modified protein Antibody, 4-Hydroxy-2-hexenal-modified , 4-Hydroxy-2-hexenal Antibody, 4-hydroxy Hexenal Antibody, HHE Antibody, 4-HHE Antibody |
| Research Areas | Cancer, Lipid peroxidation, Oxidative Stress |
| Scientific Background | Lipid peroxidation occurs when oxidizing agents attack carbon-carbon double bonds found in unsaturated lipids. In addition to membrane degradation, oxidation end-products have been found to damage cell viability through their mutagenic and toxic properties. These downstream functional consequences facilitate the development of disease and premature aging. 4-hydroxy-2-hexenal (4-HHE) is a major alpha,beta-unsaturated aldehyde product of n-3 PUFA oxidation resulting from lipid peroxidation. High levels of HHE are found in disease states (1). 4-HHE reacts with histidine residues to form Michael-addition type adducts. |
| References | 1. Long, EK., Picklo, MJ Sr. (2010) Free Radic Biol Med. 49(1):1-8. |
Product Images
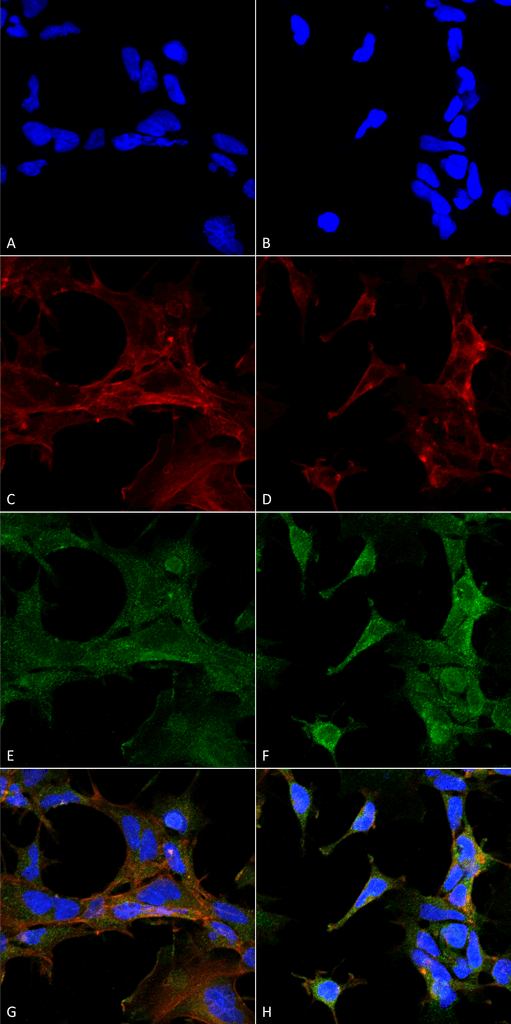
Immunocytochemistry/Immunofluorescence analysis using Mouse Anti-4-Hydroxy-2-hexenal Monoclonal Antibody, Clone 6F10 (SMC-510). Tissue: Embryonic kidney epithelial cell line (HEK293). Species: Human. Fixation: 5% Formaldehyde for 5 min. Primary Antibody: Mouse Anti-4-Hydroxy-2-hexenal Monoclonal Antibody (SMC-510) at 1:400 for 30-60 min at RT. Secondary Antibody: Goat Anti-Mouse Alexa Fluor 488 at 1:1500 for 30-60 min at RT. Counterstain: Phalloidin Alexa Fluor 633 F-Actin stain; DAPI (blue) nuclear stain at 1:250, 1:50000 for 30-60 min at RT. Magnification: 20X (2X Zoom). (A,C,E,G) – Untreated. (B,D,F,H) – Cells cultured overnight with 50 µM H2O2. (A,B) DAPI (blue) nuclear stain. (C,D) Phalloidin Alexa Fluor 633 F-Actin stain. (E,F) 4-Hydroxy-2-hexenal Antibody. (G,H) Composite. Courtesy of: Dr. Robert Burke, University of Victoria.
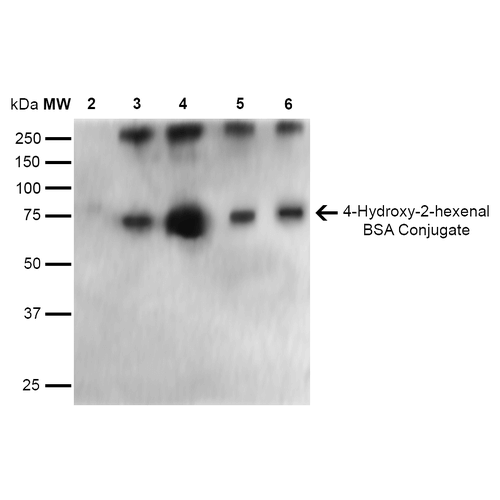
Western Blot analysis of 4-hydroxy-2-hexanal-BSA Conjugate showing detection of 67 kDa 4-hydroxy-2-hexenal protein using Mouse Anti-4-hydroxy-2-hexenal Monoclonal Antibody, Clone 6F10 (SMC-510). Lane 1: Molecular Weight Ladder (MW). Lane 2: BSA (0.5 µg). Lane 3: 4-hydroxyl nonenal-BSA (0.5 µg). Lane 4: 4-hydroxy nonenal-BSA (2.0 µg). Lane 5: 4-hydroxy-2-hexenal (0.5 µg). Lane 6: 4-hydroxy-2-hexenal (2.0 µg). Block: 5% Skim Milk in TBST. Primary Antibody: Mouse Anti-4-hydroxy-2-hexenal Monoclonal Antibody (SMC-510) at 1:1000 for 2 hours at RT. Secondary Antibody: Goat Anti-Mouse IgG: HRP at 1:2000 for 60 min at RT. Color Development: ECL solution for 5 min in RT. Predicted/Observed Size: 67 kDa.
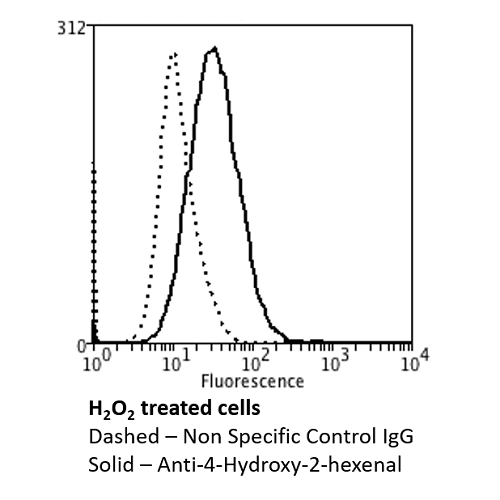
Flow Cytometry analysis using Mouse Anti-4-hydroxy-2-hexenal Monoclonal Antibody, Clone 6F10 (SMC-510). Tissue: Neuroblastoma cells (SH-SY5Y). Species: Human. Fixation: 90% Methanol. Primary Antibody: Mouse Anti-4-hydroxy-2-hexenal Monoclonal Antibody (SMC-510) at 1:50 for 30 min on ice. Secondary Antibody: Goat Anti-Mouse: PE at 1:100 for 20 min at RT. Isotype Control: Non Specific IgG. Cells were subject to oxidative stress by treating with 250 µM H2O2 for 24 hours.

![Mouse Anti-4-Hydroxy-2-hexenal Antibody [6F10] used in Western Blot (WB) on 4-hydroxy-2-hexanal-BSA Conjugate (SMC-510)](https://www.stressmarq.com/wp-content/uploads/SMC-510_4-hydroxy-2-hexenal_Antibody_6F10_WB_4-hydroxy-2-hexanal-BSA-Conjugate_1-100x100.png)
![Mouse Anti-4-Hydroxy-2-hexenal Antibody [6F10] used in Flow Cytometry (FCM) on Human Neuroblastoma cells (SH-SY5Y) (SMC-510)](https://www.stressmarq.com/wp-content/uploads/SMC-510_4-hydroxy-2-hexenal_Antibody_6F10_FCM_Human_Neuroblastoma-cells-SH-SY5Y_1-100x100.png)




















Reviews
There are no reviews yet.