Properties
| Storage Buffer | PBS pH 7.4, 50% glycerol, 0.09% Sodium azide *Storage buffer may change when conjugated |
| Storage Temperature | -20ºC, Conjugated antibodies should be stored according to the product label |
| Shipping Temperature | Blue Ice or 4ºC |
| Purification | Protein G Purified |
| Clonality | Monoclonal |
| Clone Number | 11E3 |
| Isotype | IgG1 |
| Specificity | Specific for Malondialdehyde conjugated proteins. Does not detect free Malondialdehyde. Does not cross-react with Acrolein, Crotonaldehyde, Hexanoyl Lysine, 4-Hydroxy-2-hexenal, 4-Hydroxy nonenal, or Methylglyoxal modified proteins. |
| Cite This Product | StressMarq Biosciences Cat# SMC-515, RRID: AB_2702909 |
| Certificate of Analysis | A 1:1000 dilution of SMC-515 was sufficient for detection of Malondialdehyde in 2 µg of Malondialdehyde conjugated to BSA by ECL immunoblot analysis using Goat Anti-Mouse IgG:HRP as the secondary Antibody. |
Biological Description
| Alternative Names | Malondialdehyde Antibody, MDA Antibody, Malondialdehyde (MDA) Antibody, Malonic aldehyde Antibody Propanedial Antibody, 1,3-Propanedial Antibody, Malonaldehyde Antibody |
| Research Areas | Alzheimer's Disease, Cancer, Lipid peroxidation, Neurodegeneration, Neuroscience, Oxidative Stress |
| Scientific Background | Malondialdehyde (MDA) is the biomarker in greatest diagnostic use, due to its molecular stability. This three-carbon, low-molecular weight aldehyde has a strong affinity for amino acids, which results in adduct formation to both free amino acids and proteins. Increased MDA levels have been found at correlating levels in breast cancer, and lung cancer patients. Other diseased states with elevated MDA levels include diabetes and Alzheimer’s disease. Multiple laboratory techniques exist for quantification of MDA levels, including the thiobarbituric acid reactive substances (TBARS) assay. In addition to use as a biomarker, MDA has been shown to have mutagenic effects on tissues themselves as adduct formation can result in DNA cross-linking. |
Product Images

Immunohistochemistry analysis using Mouse Anti-Malondialdehyde Monoclonal Antibody, Clone 11E3 (SMC-515). Tissue: Kidney. Species: Mouse. Primary Antibody: Mouse Anti-Malondialdehyde Monoclonal Antibody (SMC-515) at 1:100 for Overnight at 4C, then 30 min at 37C. Secondary Antibody: Goat Anti-Mouse IgG (H+L): FITC for 45 min at 37C. Counterstain: DAPI for 3 min at RT. Magnification: 10X.
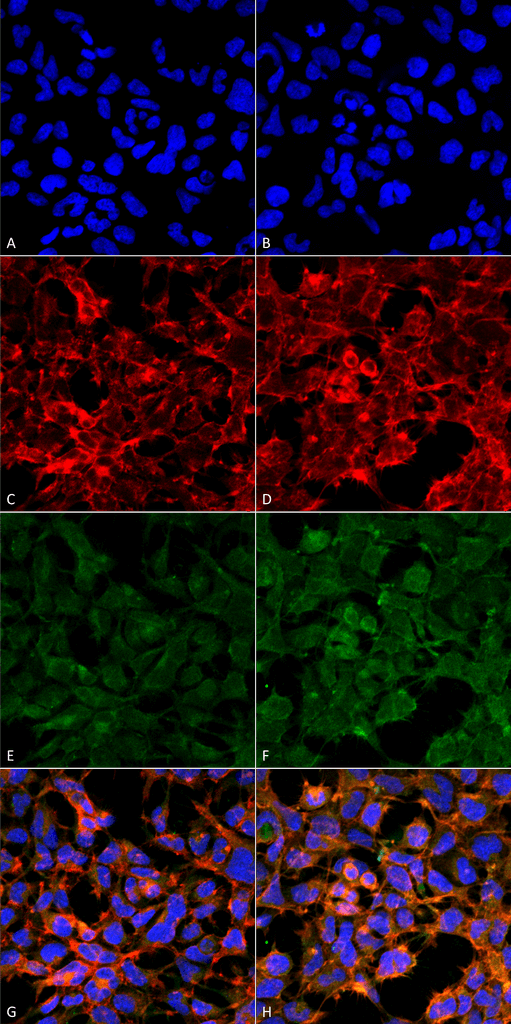
Immunocytochemistry/Immunofluorescence analysis using Mouse Anti-Malondialdehyde Monoclonal Antibody, Clone 11E3 (SMC-515). Tissue: Embryonic kidney epithelial cell line (HEK293). Species: Human. Fixation: 5% Formaldehyde for 5 min. Primary Antibody: Mouse Anti-Malondialdehyde Monoclonal Antibody (SMC-515) at 1:50 for 30-60 min at RT. Secondary Antibody: Goat Anti-Mouse Alexa Fluor 488 at 1:1500 for 30-60 min at RT. Counterstain: Phalloidin Alexa Fluor 633 F-Actin stain; DAPI (blue) nuclear stain at 1:250, 1:50000 for 30-60 min at RT. Magnification: 20X (2X Zoom). (A,C,E,G) – Untreated. (B,D,F,H) – Cells cultured overnight with 50 µM H2O2. (A,B) DAPI (blue) nuclear stain. (C,D) Phalloidin Alexa Fluor 633 F-Actin stain. (E,F) Malondialdehyde Antibody. (G,H) Composite. Courtesy of: Dr. Robert Burke, University of Victoria.
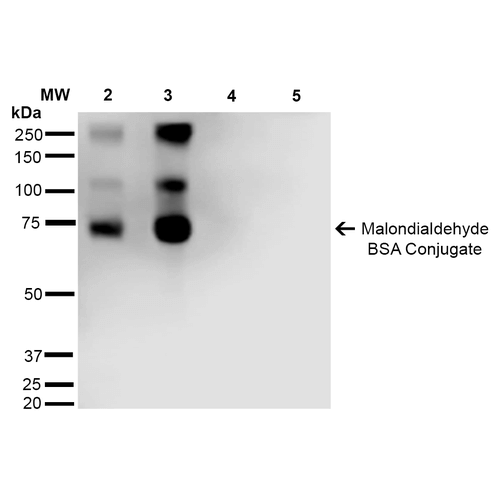
Western Blot analysis of Malondialdehyde-BSA Conjugate showing detection of 67 kDa Malondialdehyde protein using Mouse Anti-Malondialdehyde Monoclonal Antibody, Clone 11E3 (SMC-515). Lane 1: Molecular Weight Ladder (MW). Lane 2: Malondialdehyde-BSA (0.5 µg). Lane 3: Malondialdehyde-BSA (2.0 µg). Lane 4: BSA (0.5 µg). Lane 5: BSA (2.0 µg) . Block: 5% Skim Milk in TBST. Primary Antibody: Mouse Anti-Malondialdehyde Monoclonal Antibody (SMC-515) at 1:1000 for 2 hours at RT. Secondary Antibody: Goat Anti-Mouse IgG: HRP at 1:2000 for 60 min at RT. Color Development: ECL solution for 5 min in RT. Predicted/Observed Size: 67 kDa.
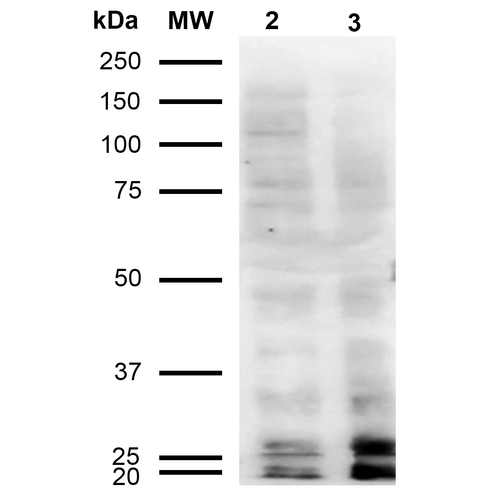
Western Blot analysis of Human Cervical cancer cell line (HeLa) lysate showing detection of Malondialdehyde protein using Mouse Anti-Malondialdehyde Monoclonal Antibody, Clone 11E3 (SMC-515). Lane 1: Molecular Weight Ladder (MW). Lane 2: HeLa cell lysate. Lane 3: H2O2 treated HeLa cell lysate. Load: 12 µg. Block: 5% Skim Milk in TBST. Primary Antibody: Mouse Anti-Malondialdehyde Monoclonal Antibody (SMC-515) at 1:1000 for 2 hours at RT. Secondary Antibody: Goat Anti-Mouse IgG: HRP at 1:2000 for 60 min at RT. Color Development: ECL solution for 5 min in RT.

![Mouse Anti-Malondialdehyde Antibody [11E3] used in Immunocytochemistry/Immunofluorescence (ICC/IF) on Embryonic kidney epithelial cell line (HEK293) (SMC-515)](https://www.stressmarq.com/wp-content/uploads/SMC-515_Malondialdehyde_Antibody_11E3_ICC-IF_Human_Embryonic-kidney-cells-HEK293_Composite_1-100x100.png)
![Mouse Anti-Malondialdehyde Antibody [11E3] used in Western Blot (WB) on Malondialdehyde-BSA Conjugate (SMC-515)](https://www.stressmarq.com/wp-content/uploads/SMC-515_Malondialdehyde_Antibody_11E3_WB_Malondialdehyde-BSA-Conjugate_1-100x100.png)
![Mouse Anti-Malondialdehyde Antibody [11E3] used in Western Blot (WB) on Cervical cancer cell line (HeLa) lysate (SMC-515)](https://www.stressmarq.com/wp-content/uploads/SMC-515_Malondialdehyde_Antibody_11E3_WB_Human_Cervical-Cancer-cell-line-HeLa_1-100x100.png)




















Reviews
There are no reviews yet.